Tap into a new possibility for adolescent depression!!

A New Possibility for Adolescent Depression
If you are looking for another option to treat adolescent depression, you are not alone. Each year, 1 in 5 US adolescents experience at least one major depressive episode.
The #1 Physician Recommended TMS Treatment
Proven safe and effective for adults struggling with depression since 2008, NeuroStar® has been performed over 6.1 million times, in more than 169,000 patients.
Nearly 10,000 adolescents have been treated with over 300,000 NeuroStar sessions.

Proven Effective for Adolescents
NeuroStar is a non-drug, non-invasive TMS treatment DFA-cleared for adolescents, as an adjunct (add-on) to your existing therapy for ages 15 an older.
With NeuroStar, All You Have to Do is Show Up
- A session lasts as little as 19 minutes per day
- Resume normal activities immediately after treatment
- 36 treatment sessions to completion
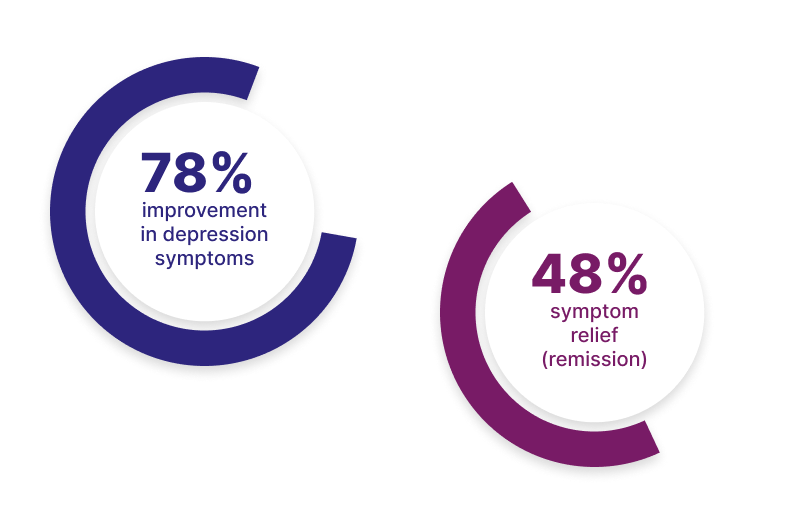
1. FDA Clearance K231926 The outcomes reported represent the subset of study patients for which the CGI-S data was reported before and after an acute course of NeuroStar TMS. Patients aged 12 to 21 (average 19.2 ± 1.5) were treated under real-world conditions where patients may have been prescribed concomitant depression treatments including medications. “Measurable relief” was defined as a CGI-S score ≤3 and “complete remission” was defined as a CGI-S score ≤2 at the end of treatment.
2. National Institute of Mental Health. https://www.nimh.nih.gov/ health/statistics/major-depression#part_2565.
3. Post A, et al. J Psychiatric Research. 2001; 35:193-215.
4. Liston C, Chen AC, Zebley BD, et al. Biol Psychiatry. 2014; 75(7);517-526.
5. Data on file. Neuronetics, Inc. 2024.
Important Safety Information:
The NeuroStar Advanced Therapy System is indicated as an adjunct for the treatment of Major Depressive Disorder (MDD) in adolescent patients (15-21 years old).
NeuroStar Advanced Therapy is only available by prescription. A doctor can help decide if NeuroStar Advanced Therapy is right for you. Patients’ results may vary.
The most common side effect is pain or discomfort at or near the treatment site. These events are transient; they occur during the TMS treatment course and do not occur for most patients after the first week of treatment. There is a rare risk of seizure associated with the use of TMS therapy (<0.1% per patient).
